NEW YORK/GENEVA, JULY 28, 2020—Cepheid, a US company that produces rapid, point-of care COVID-19 tests, is prioritizing high-income countries at the expense of people in low resource settings, said Doctors Without Borders/Médecins Sans Frontières (MSF) today. The international medical humanitarian organization called on Cepheid to allocate these urgently needed COVID-19 tests equitably and affordably across all countries.
“As countries are struggling to deal with suspected COVID-19 cases, having an accurate rapid diagnostic test is essential for real-time management of people affected with the virus in order to tackle this pandemic,” said Dr. Greg Elder, medical coordinator for MSF's Access Campaign. “So many lives could be saved if corporations like Cepheid made their tests available urgently and affordably in all countries.”
MSF is also calling on Cepheid to refrain from profiteering off of the pandemic and lower the price of each test, called Xpert Xpress SARS-COV2, from the nearly $20 it currently charges to $5.
Cepheid—which developed the COVID-19 test with $3.7 million in public funding from the United States government’s Biomedical Advanced Research and Development Authority, an office under the Department of Health and Human Services—has currently set the price for each test at $19.80 in 145 developing countries, including the world’s poorest countries where people live on less than two dollars per day.
“It is indefensible for Cepheid to profit off this pandemic,” said Sharonann Lynch, senior HIV & TB advisor for MSF’s Access Campaign. “This is not the time to set the price based on what the market can bear. This critical test must be made accessible to all people who urgently need it at $5 per test to tackle this global health emergency.”
MSF and others’ analysis of the cost of manufacturing Cepheid’s tuberculosis (TB) test for which the company charges $10 in developing countries—a test similar to the one for COVID-19—shows that the cost of goods, including materials, manufacturing, overhead, and other indirect expenses for each test is as low as $3 at high volumes and with relevant royalties expired. Therefore, each test could be sold at a profit for $5.
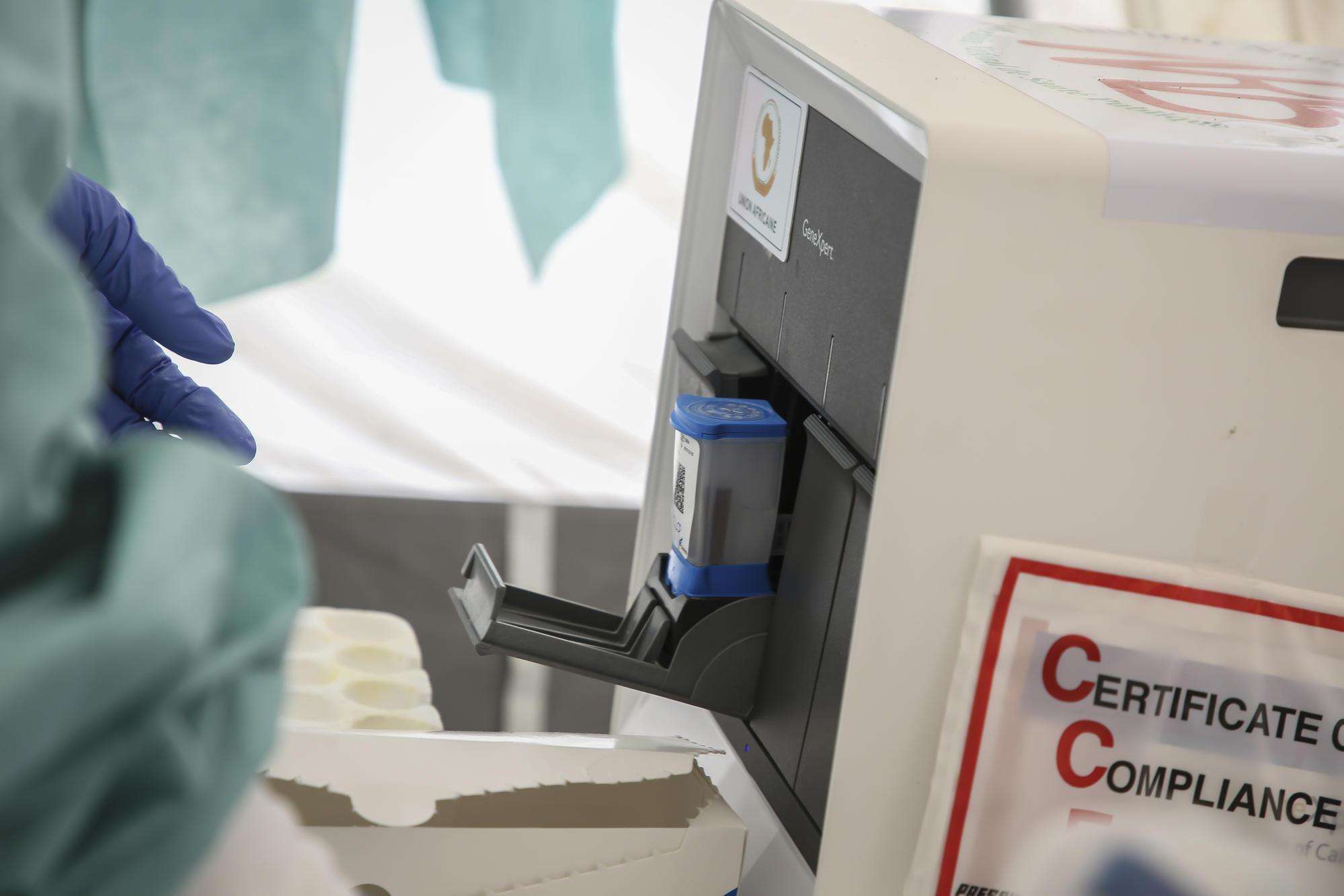
The test is designed for use with Cepheid’s GeneXpert testing platforms, which are already being used across the world to diagnose TB, Ebola, and other infectious diseases. There are an estimated 11,000 GeneXpert machines in low- and middle-income countries that could run COVID-19 tests if the Xpert Xpress SARS-COV2 test cartridges were widely and affordably available.
But it’s not just price; Cepheid must commit to making these tests available in low- and middle-income countries.
The World Health Organization “Diagnostics Consortium” was set up in March 2020 to support rapid and equitable access to COVID-19 health products and diagnostics for low- and middle-income countries. While the Consortium secured volume commitments from major diagnostic test manufacturers—Abbott, Cepheid, Roche, and Thermo Fisher—for a period of four months, the number of tests committed from Cepheid represented only one-third of its COVID-19 test manufacturing capacity. As a result, less than half of countries’ orders into the Consortium have been fulfilled.
“In this raging pandemic, wealthier countries have a tremendous advantage over others in buying COVID-19 medical tools for their use first,” Lynch said. “We are deeply concerned that people in many resource-limited countries will be deprived of this critical diagnostic test.”
The Consortium and corporations, including Cepheid, are set to meet again in the coming weeks to negotiate supply volumes and prices for the next four-month period from September-December.
“We need to see Cepheid take the right steps and guarantee a fair allocation and affordable supply of its COVID-19 test to the Diagnostic Consortium to help countries that would otherwise be left behind or left out of bilateral deals,” Lynch said. “No one should be denied access based on where they come from or what they earn.”
MSF teams are racing to respond to the COVID-19 pandemic in dozens of countries by adapting existing activities to the COVID-19 pandemic as well as opening new projects. MSF’s response focuses on three main priorities: supporting authorities to provide care for COVID-19 patients; protecting people who are vulnerable and at risk; and keeping essential medical services running.
Cepheid charges four times more than it should per COVID-19 test
Pharmaceutical corporation overcharges and undersupplies WHO effort to test people for COVID-19 in developing countries
Democratic Republic of Congo 2018 © Carl Theunis/MSF



